The heart is a complicated electrical system that generates electrical signals and runs the pumps. It causes them to contract and relax in a specific timing sequence. It either speeds up or slows down the electrical impulses responsible for each heartbeat.
If the heart beats too slowly, it will not pump enough blood to meet the body’s demands. If the heart beats too quickly, it will not have enough time between contractions to fill and will fail to pump enough blood to the body. As an essential part of the body, what is the origin of the heart?
It goes back at least 520 million years when an underwater landslide in ancient China buried a tiny arthropod called Fuxianhuia protensa. Fuxianhuia was small, no bigger than one of the credit cards. With a segmented body, an impressive pair of mandibles, and eyes on the ends of two stalks.
It had the tools of a swift ocean predator, so it probably needed a lot of oxygen. It may explain why Fuxianhuia is the earliest animal known to have had a blood circulatory system and an organ to keep its blood moving: the first known traces of a very early heart.
The path that connects this ancient predator is long, with many side tracks leading to other, more familiar organisms with hearts. It includes animals like slugs, snails, insects, and crustaceans. The first vertebrates to walk on four legs. It took hundreds of millions of years and countless different iterations of the same basic structure to lead to the heart.
How did the heart evolve?
The history of the heart’s origin is as old as animal life. To understand where hearts came from, we must return to the earliest common ancestor of everything with a heart. Scientists don’t have a fossil of that ancestor, but they have some proof of its existence: its genes. The heart carries some of the same genes that this organism has. Those genes are responsible for developing blood vessels that can contract to push blood around.
Here is a general overview of the evolution of the heart:
Simple Pumping Mechanisms: The earliest ancestors of animals had simple, tubular structures that functioned as pumps to circulate fluids. These structures gradually evolved into more specialized organs over time.
Evolution of Chambered Hearts: In some invertebrate groups, such as mollusks and arthropods, the heart evolved into a more complex organ with multiple chambers. These early chambered hearts enabled the more efficient circulation of oxygen and nutrients to the tissues.
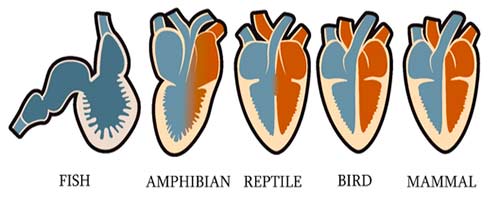
Evolution of Vertebrate Hearts: The evolution of the vertebrate heart involves further modifications and adaptations. The earliest vertebrates had two-chambered hearts with a single atrium and a single ventricle, capable of pumping blood in a single loop through the gills and back to the body.
Development of Three and Four-Chambered Hearts: Over time, vertebrates developed more advanced cardiovascular systems. The evolution of a partial or complete separation of the ventricle into two chambers resulted in three-chambered hearts found in amphibians and reptiles. Later, some reptiles and birds evolved four-chambered hearts, completely separating oxygenated and deoxygenated blood, enabling more efficient oxygen delivery to the body.
Mammalian Hearts: Mammals, including humans, have four-chambered hearts. The mammalian heart has a highly specialized structure with two atria and two ventricles, completely separating oxygenated and deoxygenated blood. This adaptation allows for a more efficient oxygen supply to the body’s tissues.
Now the question is, how was the heart shape created? Over time, an incredibly diverse group of animals has used this basic set of instructions to arrive at different solutions for circulating blood. Researchers can tease apart the history of the heart by studying the genomes of living organisms that share these heart-making genes.
Using that method is known as the molecular clock. It combines genetic mutation rates with insights from well-dated fossils. The common ancestor of all animals with a heart lived in the Neoproterozoic Era, between 600 million and 700 million years ago. Even though it gave rise to all organisms with hearts, this ancestor probably didn’t have a heart of its own.
So the genes that form hearts today are older than the heart itself! Instead of a heart, researchers believe that this ancestor used blood vessels to push blood through its body. This type of system is called a peristaltic pump. It works like the intestines, with muscles gradually squeezing to move material. Annelid worms and the marine chordates, known as lancelets, have held on to this simple model of blood circulation.
So scientists think this is what ancestor had the simplest version of a system shared by all its descendants. Over time, practically every animal descended from that ancestor has added complexity to this basic system. They did so independently, as natural selection repeatedly acted on that peristaltic pump and the genes that make it possible. The circulatory systems in these animals often became more complex because they tended toward more active lifestyles.
For worms, a peristaltic pump works fine. But that wasn’t effective enough for active predators and animals that need to move quickly. The solution for most animals was a heart supplying more complex and active bodies with more oxygen. Not all animals today have a genuine heart. But some version of an organ for circulating blood has evolved in at least three major groups:
- Arthropods.
- Mollusks.
- Vertebrates.
Arthropods heart
The arthropod version of a “heart” is a long, tube-like structure that runs the length of the animal’s body and is called a dorsal vessel. Its job is to collect blood and propel it in one direction: toward the head. It doesn’t look like a heart; not all researchers will call it one. But it performs a comparable function. That dorsal vessel has been around for a long time.
In 2014, researchers studied a specimen so well preserved that they could make out traces of its dorsal vessel. The scientists reconstructed it based on its similarities with the hearts of modern arthropods. So Fuxianhuia had this dorsal vessel, but it’s unclear when this structure first evolved in arthropods.
Researchers still have a healthy debate about when different lineages gained or lost their blood-pumping organs. Because different kinds of arthropods have dorsal vessels that differ from each other, some don’t have any at all! So this makes it hard to say if these vessels are a single ancestral feature or something that showed up on its own, convergently, many times.
Mollusks heart
This group includes gastropods (snails and slugs), cephalopods (squid and octopodes), and bivalves (clams and mussels), among many others. All of these animals have hearts. So it’s likely that their common ancestor had a heart too.
The ancestor of all mollusks goes even further back in the Ediacaran Period, to a fossil of a squishy tiny seafloor dweller known as Kimberella. This animal is sometimes interpreted as a mollusk. If it is one, then the common ancestor of mollusks. Its ancestral heart has to be as old as it and the other earliest fossil mollusks. That’s at least 550 million years old.
Unlike arthropods, mollusks have hearts with multiple chambers. Generally, one or more chambers collect oxygenated blood from the gills. These are called auricles. Then they feed the oxygen-rich blood into another chamber called a ventricle. It pumps the blood out to the rest of the body. The more complex the mollusk, the more complex its heart is.
For example, in addition to a central chambered heart, most cephalopods like cuttlefish, octopodes, and squid have two extra branchial hearts that pump blood to their gills. Three hearts might seem like a lot. But they may have played a key role in helping some cephalopods develop a more active lifestyle.
Vertebrates heart
Vertebrates have also evolved more complex hearts. Molecular clock studies suggest that some genes responsible for creating heart muscle evolved in vertebrates from 540 million to 570 million years ago. Meanwhile, the genes that make the specialized tissue that lines blood vessels, known as the endothelium, date back between 510 million to 540 million years ago.
By the end of the Ediacaran Period, the vertebrate circulatory system was well on its way. The earliest fossil of a vertebrate heart belongs to a fish called Rhacolepis. Also, it has two chambers, as all fish hearts do. But Rhacolepis lived only about 115 million years ago.
The hearts of the earliest vertebrate ancestors probably were like that found in Rhacolepis. It likely had two chambers: an atrium to collect blood and a ventricle to push it back to the body. That’s how the hearts of vertebrates start during embryonic development. All vertebrate hearts develop according to a common pattern:
- In the embryo, a blood vessel twists in on itself to create two chambers. Then it develops depending on what kind of organism it is and other types of genetic instructions it has.
Every vertebrate develops a heart that suggests that a two-chambered plan is shared. Again, vertebrate hearts became more sophisticated as the animals became more complex and active. Amphibians, for example, first appeared in the fossil record around 360 million years ago; today, most have two atria.
- One for collecting oxygenated blood and one for collecting deoxygenated blood, as well as a single ventricle.
Some more basal or primitive living amphibians have chambers that aren’t separated. Sort of like they have two and a half chambers. So their common ancestor probably didn’t have three well-defined chambers, either, and instead, that came later.
- Mammals have four chambers; although shared, mammalian ancestors appeared around 200 million years ago.
- In the mammalian heart, the right atrium and ventricle collect deoxygenated blood and send it to the lungs. The left atrium and ventricle collect the oxygenated blood from the lungs and send it to the body.
So, arthropods, mollusks, and vertebrates acquired some version of a heart within the same general stretch of geologic time. But even though all of these hearts look quite different, they’re all variations on a theme derived from the same basic set of genetic instructions.
In 1993, a researcher in Michigan found that the dorsal vessel in fruit flies develops with the help of a gene known as Tinman. Without a Tinman gene, the flies don’t have a heart? This gene instructs developing tissues to differentiate into various kinds of cardiac cells.
Later studies found that vertebrates have the equivalent of the Tinman gene. It turns out that our gene is similar enough to Tinman’s. Researchers believe they evolved from a single, earlier gene in that common ancestor that lived more than 600 million years ago.
In humans, this gene is given the less-punny name of Nkx2-5. It instructs the developing tissue. To generate the many different kinds of cells that make a heart. However, the dorsal vessel of a fly and the present heart doesn’t seem to have much in common with 600-million-year-old ancestors.
Over vast stretches of geologic time, natural selection has acted on this genetic legacy over and over as animals became more complex. The result is a variety of fruit flies, cuttlefish, and Fuxianhuia, a humble arthropod from the distant past.
More Articles:
Full Process Of Blood Transfusions
What Makes Horseshoe Crab Blood Expensive?
How Did Plants Convert Into Carnivorous?
References:
RM Alexander, The Collins Encyclopedia of Animal Biology.
Anderson RH, Ho SY, Redmann K, The anatomical arrangement of the myocardial cells making up the ventricular mass.
Axelsson M, Franklin, From anatomy to angioscopy: 164 Years of crocodilian cardiovascular research, recent advances, and speculations.
Bajaj P, Tang X, Saif TA, Stiffness of the substrate influences the phenotype of embryonic chicken cardiac myocytes.
Bardack D, First fossil hagfish (myxinoidea), a record from the Pennsylvanian of Illinois.
Exploring the Expression of Cardiac Regulators in a Vertebrate Extremophile: The Cichlid Fish Oreochromis (Alcolapia) alcalica.
Automated selection of homologs to track the evolutionary history of proteins.
The Memory of the Heart, Cirillo M.
Scoville, Heather. “Evolution of the Human Heart’s Four Chambers.”
Table of Contents
