Enzymes are nature’s catalysts, orchestrating the complex symphony of life’s processes with precision and efficiency. They are the unsung heroes in the microscopic world, facilitating almost every chemical reaction in biological systems, accelerating them to a pace that sustains life.
Enzymes are biological molecules that act as catalysts, speeding up chemical reactions in living organisms by lowering the activation energy required for these reactions to occur. They achieve this by binding to specific substrates, facilitating their transformation into products, and remaining unchanged and available for further reactions.
This exploration focuses on the fascinating mechanics of how enzymes work, revealing the elegance of their function and their indispensable role in maintaining the delicate balance of our biological existence.
How Do Enzymes Work?
Enzymes are proteins that act as biological catalysts, facilitating chemical reactions in living organisms. They work by lowering the activation energy required for a reaction to occur. Here’s a general overview of how enzymes work:
Substrate Binding: Enzymes have a specific three-dimensional structure with a region called the active site. The substrate, the molecule or molecules upon which the enzyme acts, binds to the active site through precise molecular interactions such as hydrogen bonding, electrostatic interactions, and hydrophobic interactions.
Enzyme-Substrate Complex Formation: When the substrate binds to the active site, it forms an enzyme-substrate complex. This complex is stabilized by weak interactions, allowing the enzyme to hold the substrate in a specific orientation conducive to the chemical reaction.
Catalytic Reaction: Once the enzyme-substrate complex is formed, the enzyme can catalyze the conversion of the substrate into the desired products. Enzymes can accelerate reactions by providing an alternative reaction pathway with lower activation energy. They achieve this through several mechanisms:
a. Strain or Stress: Enzymes can induce strain on the bonds within the substrate, making them more reactive and prone to breaking or forming new bonds.
b. Proximity and Orientation: Enzymes bring the reacting molecules (substrates) close together, increasing the chance of successful collision and reaction. They also orient the substrates in a specific configuration that favors the formation of the desired products.
c. Active Site Chemistry: The active site of an enzyme contains specific amino acid residues that participate in the chemical reaction. These residues can act as acids, bases, or catalysts, facilitating the transfer of protons or electrons between the substrate molecules, stabilizing reaction intermediates, or promoting specific chemical transformations.
Product Formation and Release: After the catalytic reaction, the enzyme facilitates the release of the products. The products have a lower affinity for the active site than the substrate, allowing them to be released, thus regenerating the active enzyme for further catalysis.
Enzyme Regulation: Enzyme activity can be regulated to meet the organism’s needs. This regulation can occur through various mechanisms, such as feedback inhibition, allosteric regulation, covalent modification, or changes in enzyme concentration.
Like all other catalyst enzymes, almost every biochemical reaction is catalyzed by an enzyme, increasing chemical reactions without being consumed or permanently altered. However, they differ from ordinary chemical catalysts in:
- Higher catalytic power (Higher reaction rates).
- Greater reaction specificity.
- Milder reaction conditions.
- Capacity for regulation.
Few non-biological catalysts have all these properties. However, the catalytic mechanism employed by enzymes is identical to those used by chemical catalysts. Enzymes are better designers and are biologically relevant conditions, and catalyze reactions are slow.
Mechanism of enzyme action: Lock & Key Hypothesis
What are the lock and key hypotheses? It is related to the relationship between substrate and enzyme. The substrate can be referred to as a key. The enzyme’s active site can be called lock and thus key and lock mechanisms.
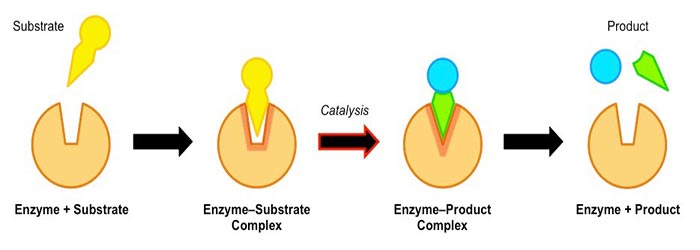
It indicates that the substrate has a complementary shape with the enzymes’ active site. It means only a specific substrate can fit it. They are specific reactions because only a particular substrate can bind to the active site. Not any substrate can bind to any active site.
The lock and key model is the induced fit model that describes how our binding occurs more correctly. The substrate fits precisely and correctly into the active side to complement their complementary shapes in the lock and key model.
It moves into the active side when it fits, forming those non-covalent interactions. Our induced fit model shows that the enzyme’s active site does not complement our substrate. Still, when the binding takes place, the enzyme conforms to the structure of that substrate. So, the enzyme’s active site changes shape ever so slightly.
The enzyme’s active site shape is not exactly complimentary with the induced fit model. However, upon binding the substrate to the active site, the binding causes the active site to complement the substrates. The induced-fit model tells us it is when the binding occurs at the active site of that enzyme.
So, the substrate becomes complementary to the active side, and the active site becomes complementary to that particular substrate. The induced-fit model correctly describes the binding between the enzyme and the substrate’s active site.
The catalytic activity of enzymes involves their binding or substrates to form an enzyme-substrate complex. The substrate binds to a specific enzyme region called its active site. The substrate is converted into the reaction product, releasing it from the enzyme.
A peak denotes the transition state. The difference between the ground state’s energy levels and the transition state is called Gibbs free activation energy. Or simply the activation energy Delta G denotes it.
Here are 5 methods to describe the mechanism of the enzyme.
Non-covalent interactions
Let us study non-covalent interactions between enzyme and substrate, like non-covalent bonds, hydrogen bonds, hydrophobic and ionic interactions. These interactions are accompanied by a release of free energy called binding energy.
This binding energy contributes to specificity as well as to catalysis. This binding energy ultimately derives much of the catalytic power of enzymes. As it is a significant source of free energy used by enzymes to lower the activation energies of reactions as per the equation,
V= k [S] = kT/h [S] e^-∆G/RT
About 5.7 kilojoules per mole must lower g to accelerate the first-order reaction by a factor of 10 under conditions commonly found in cells. The energy from forming a weak interaction is generally estimated to be 4 to 30 kilojoules per mole.
Therefore, the overall binding energy levels of many such interactions are sufficient to lower activation energies by 6,200 kilojoules per mole. The same binding energy that provides energy for catalysis also gives an enzyme.
We have all these different types of enzymes found inside our bodies. They decrease the reaction’s activation energy, but how exactly is that achieved, and what are some mechanisms? What are some enzymes’ methods to achieve this decrease in activation energy?
Covalent catalysis
Many enzymes contain active sites with catalytic residues that form covalent bonds with the substrate molecule. They also keep that molecule in place for the time being until that reaction takes place.
The enzyme is never used or, depleted, or changed in any reaction. We have to break that bond, and that’s exactly why we call this bond a temporary or a transient covalent bond. For example, some enzymes include trypsin, chymotrypsin, and other digestive enzymes.
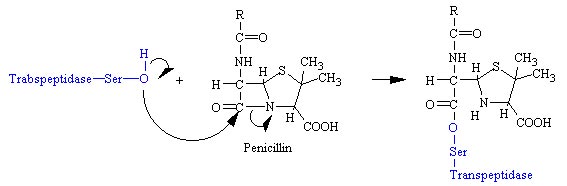
In the first step of this reaction, this molecule forms a bond between the oxygen and this carbon, kicking off this terminal amino acid to form the following temporary, transient acyl intermediate molecule. At the end of the reaction, this bond is broken.
It forms the bond to keep this group attached to the active site so that another substrate can move in and grab this group. So, the bacterial enzyme glycol peptide transpeptidase utilizes covalent catalysis. Chymotrypsin is an important digestive enzyme that exists inside the digestive system.
Catalysis by proximity
By collision theory, two substrate molecules about to react must collide. They must collide with enough energy and with the proper orientation.
When we form the product molecule when the collision occurs with the proper orientation and the right amount of energy, do we form a product molecule? They bring the substrate molecules into the tiny space, creating a microenvironment for that reaction.
So, inside the active site, they create a microenvironment that brings those substrate molecules nearby but also orients those subject molecules in the proper orientation.
- Many biological reactions involve two or more substrate molecules. It implies that for a reaction to take place. They must be close enough and must also have the proper orientation.
- Active sites provide a microenvironment that brings the substrate close enough for collisions to occur at a high frequency. The active sites may also orient the molecules properly for that bond to form and form those products.
Acid-base catalysis
Many residues are involved, or specific residues are found in active sites that transfer an H ion. One specific residue is the histidine amino acid. So, the histidine molecule has a pH relatively close to the normal physiological pH. Active sites may contain residues such as histidine that can transfer hydrogen ions. If a hydrogen ion transfers from one molecule to another, it creates a strong nucleophile. That strong nucleophile might be needed in that particular biological reaction.
The active site may activate a nucleophile required in that catalysis by transferring the hydrogen ion. Also, It can stabilize different groups that might be found inside the activities containing charges. The transfer of hydrogen ions can increase the electrostatic interactions within that active site. It can, in turn, stabilize things like the transition state inside that chemical reaction.
Example: One particular example of an enzyme that uses acid-base catalysis is chymotrypsin. Inside the chymotrypsin active site, serine residue acts as a nucleophile.
The hydrogen ion from the oxygen of serine must be taken away. The hydrogen atom is transferred onto the nitrogen. The histidine side chain delocalizes the positive charge among these different atoms. But this one now contains a full negative charge.
Metal-ion catalysis
The mechanism by which enzymes can decrease the activation energy and increase reaction rates is called metal ion catalysis. Example: myoglobin and hemoglobin. These proteins use metal atoms, and enzymes utilize metal as cofactors.
What’s so special about these metal atoms? Metal atoms can lose electrons very quickly, and by losing electrons, they gain a positive charge. So, they are deficient in electrons. They have a positive charge, interacting with molecules inside the active site. They can stabilize the transition states and the intermediate molecules formed within that active site.
Example: A zinc metal atom is used to form a strong nucleophile. The hydroxide nucleophile and metal atom can hold that substrate molecule in place.
So, in the same way, we can use covalent catalysis to orient that substrate and hold it in place. We can also use the positive charge of these metal atoms to bring the substrate molecules in the proper orientation and hold them inside the active site so that the reaction can occur at a reasonably high rate.
Enzymes are the biological catalysts that speed up the rates of all reactions inside our cells. I hope you will understand the working principle and mechanism of enzymes properly. If you have any questions, please ask me in the comment section.
How do enzymes lower activation energy?
Enzymes are fascinating biological catalysts that play a crucial role in speeding up reactions by lowering the activation energy required for the reaction to occur. Activation energy is the energy barrier that must be overcome for reactants to be converted into products in a chemical reaction. Here’s a detailed explanation of how enzymes achieve this:
Substrate Binding: Enzymes have a specific region called the active site, where substrates—the molecules upon which enzymes act—bind. The unique shape of the active site matches the shape of its substrate(s), following the lock-and-key model, or adjusts to fit the substrate, according to the induced fit model. This specificity ensures that enzymes interact only with particular substrates.
Transition State Stabilization: The key to lowering activation energy lies in the enzyme’s ability to stabilize the transition state, which is the state where bonds in the substrates are partially broken and new ones are partially formed. By stabilizing the transition state, enzymes decrease the amount of energy required to reach this state, effectively lowering the activation energy barrier.
Optimal Orientation: Enzymes can also lower activation energy by bringing substrates together in the optimal orientation for a reaction. This reduces the entropy (disorder) of the system and increases the likelihood of a successful reaction, decreasing the total energy needed for the reaction to proceed.
Microenvironment Adjustment: Enzymes create a specific microenvironment within the active site that is more conducive to the reaction. For example, if a reaction requires a lower pH, the active site might be more acidic, facilitating proton transfer. This adjustment makes it easier for the reaction to proceed, lowering the activation energy.
The strain on Bonds: When substrates bind to the enzyme’s active site, the enzyme strains bonds in the substrate, making them easier to break. This strain helps to reach the transition state faster and with less energy input than required without the enzyme.
Temporary Covalent Bond Formation: Some enzymes work by forming a temporary covalent bond with the substrate. This makes it easier to break other bonds within the substrate or facilitate the transfer of atoms or groups within the substrate, thus lowering the activation energy required for the reaction.
The reduction of activation energy allows biochemical reactions to occur rapidly and efficiently at the relatively mild temperatures of biological systems, facilitating the complex and highly regulated metabolic pathways necessary for life. Without enzymes, these reactions would proceed too slowly to sustain life as we know it. Enzymes thus speed up reactions and enable the precise regulation of metabolic pathways, allowing organisms to respond to changes in their environment and maintain homeostasis.
How do enzymes speed up reactions?
Enzymes speed up chemical reactions without being consumed or permanently altering themselves, acting as catalysts. They achieve this remarkable feat primarily by lowering the activation energy of a reaction, making it easier for the reaction to proceed at a faster rate. Here’s a breakdown of how enzymes accelerate reactions:
Lowering Activation Energy: As mentioned, enzymes lower the activation energy required for a reaction. Activation energy is the energy barrier that must be overcome for reactants to be transformed into products. By stabilizing the transition state and reducing this barrier, enzymes allow the reaction to proceed more quickly without their presence.
Substrate Specificity and Binding: Enzymes are highly specific; each enzyme typically binds to one or a few substrates. This specificity is due to the enzyme’s active site’s unique shape and chemical properties. When the substrate binds to the active site, it forms an enzyme-substrate complex, facilitating the chemical reaction.
Creating an Optimal Environment: An enzyme’s active site binds the substrate and provides an optimal environment for the reaction. This includes a particular pH, ionic strength, or the presence of specific cofactors or coenzymes necessary for the reaction to proceed efficiently.
Bringing Substrates Together: For reactions that involve two or more substrates, enzymes bring these substrates together in the correct orientation and proximity for the reaction to occur. This reduces the entropy (disorder) of the system and increases the rate at which the reactants collide in the correct orientation, thereby speeding up the reaction.
Inducing Strain on Substrates: By binding to its substrates, an enzyme can induce strain on the bonds within those substrates, making them more reactive. This strain lowers the energy level of the substrate’s ground state, making it easier for the reaction to proceed to the transition state.
Providing an Alternative Reaction Pathway: Enzymes offer a different pathway for the reaction with a lower activation energy than the non-catalyzed reaction. This alternative pathway allows the reaction to proceed more rapidly.
Direct Participation in the Reaction: Some enzymes directly participate in the chemical reaction by forming transient covalent bonds with the substrate. This helps properly position the substrate, facilitating the transfer of electrons or groups within the substrate, thus speeding up the reaction.
Enzymes are incredibly efficient, often increasing reaction rates by millions or even billions. For example, without the enzyme catalase, the decomposition of hydrogen peroxide to water and oxygen would occur so slowly that it would be imperceptible. With catalase present, this reaction occurs rapidly, protecting cells from oxidative damage.
Through these mechanisms, enzymes play an essential role in regulating the speed of biochemical reactions, ensuring that the metabolic processes necessary for life proceed quickly enough to sustain life but not so rapidly that the cell loses control over its metabolic activities.
How do enzymes work in the body?
Here’s a detailed overview of how enzymes work in the body across various systems and processes:
- Metabolism and Digestion
Digestive Enzymes: Enzymes in the digestive system break down large, complex food molecules into smaller, absorbable ones. For example, amylase breaks down starches into sugars in the mouth and small intestine, while proteases break down proteins into amino acids in the stomach and small intestine. Lipases break down fats into fatty acids and glycerol in the small intestine.
Metabolic Enzymes: Within cells, enzymes are key to metabolic pathways that convert nutrients into energy and new materials for cellular growth, repair, and maintenance. For instance, enzymes in the citric acid (Krebs) cycle in mitochondria help generate ATP, the cell’s energy currency, from glucose and other nutrients.
- DNA Replication, Repair, and Transcription
DNA Polymerases: These enzymes are crucial for DNA replication, allowing cells to duplicate their genomes before cell division.
Repair Enzymes: They identify and correct damaged or mismatched DNA, ensuring genetic information is accurately maintained and transmitted.
RNA Polymerases: Enzymes involved in transcribing DNA into RNA, a key step in gene expression and the synthesis of proteins.
- Protein Synthesis and Modification
Ribozymes: A type of RNA that acts as an enzyme, playing a critical role in various RNA processing reactions, including RNA splicing and translation.
Transferases: Enzymes that catalyze the addition of phosphate groups (phosphorylation) or other molecules to proteins, altering their activity, localization, or stability.
- Detoxification
Cytochrome P450 Enzymes: Located primarily in the liver, these enzymes metabolize potentially harmful compounds, including drugs and toxins, making them easier for the body to excrete.
- Energy Conversion and Storage
Kinases and Phosphatases: These enzymes play key roles in regulating cellular energy metabolism by adding or removing phosphate groups from molecules, thus activating or deactivating enzymes or signaling pathways involved in energy production and consumption.
Glycogen Synthase and Glycogen Phosphorylase: Enzymes are involved in converting glucose to glycogen for storage and back to glucose for energy, respectively.
- Immune Response and Inflammation
Lysozyme: An enzyme found in saliva, tears, and other body fluids, capable of breaking down the cell walls of certain bacteria, contributing to the body’s first line of defense against infection.
Cyclooxygenases (COX enzymes): Involved in synthesizing prostaglandins, which play a role in inflammation and pain, making them targets for anti-inflammatory drugs.
- Signal Transduction
Kinases and Phosphatases (again): These enzymes are also crucial in signaling pathways, transmitting signals from the cell’s exterior to its interior, which can affect growth, division, and death.
Enzymes are thus integral to nearly every biochemical process in the body. Their highly specific nature ensures that each reaction occurs at the right place and time, under optimal conditions, and with appropriate regulation. Without enzymes, the chemistry of life would be too slow to sustain life as we know it.
How do enzymes work in the digestive system?
Enzymes in the digestive system play a crucial role in breaking down food into smaller, absorbable components. This process allows the body to extract nutrients from food, converting them into energy and building blocks for repair and growth. Here’s a detailed look at how these specialized proteins function throughout the digestive tract:
- Mouth
Salivary Amylase (Ptyalin): The digestion process starts in the mouth, where salivary amylase begins the breakdown of starches into maltose and dextrins. The salivary glands produce this enzyme, which is active the moment food enters the mouth, initiating the chemical process of digestion alongside the mechanical process of chewing.
- Stomach
Pepsin: In the stomach, the enzyme pepsin, activated from its precursor pepsinogen in the presence of stomach acid (HCl), starts the digestion of proteins, breaking them down into smaller peptides. The acidic environment of the stomach facilitates the activation and function of pepsin.
Lipase: Although fat digestion primarily occurs in the small intestine, gastric lipase also works in the stomach to break fats into fatty acids and glycerol, albeit to a lesser extent than its intestinal counterpart.
- Small Intestine
Pancreatic Enzymes: The pancreas plays a vital role in digestion by secreting a mixture of enzymes into the small intestine through the pancreatic duct. These include:
Pancreatic Amylase: Continues the breakdown of starch into maltose and dextrins.
Trypsin and Chymotrypsin: Further break down polypeptides into shorter peptides. They are secreted as inactive precursors (trypsinogen and chymotrypsinogen) and are activated in the small intestine.
Pancreatic Lipase is the major fat-digesting enzyme that breaks down triglycerides into fatty acids and glycerol.
Ribonuclease and Deoxyribonuclease: Break down RNA and DNA into their constituent nucleotides.
Intestinal Enzymes: The lining of the small intestine produces additional enzymes that work on the surfaces of the intestinal cells to break down di- and tripeptides into amino acids, maltose into glucose, and other disaccharides into monosaccharides. These enzymes include:
Peptidases: Break down peptides into amino acids.
Sucrase, Lactase, and Maltase: Break down sucrose into glucose and fructose, lactose into glucose and galactose, and maltose into two glucose molecules, respectively.
- Absorption
After digestion by these enzymes, the resulting small molecules—amino acids, sugars, fatty acids, and glycerol—are then absorbed through the intestinal wall into the bloodstream or the lymphatic system (in the case of fats) to be used by the body for energy, growth, and repair.
Regulation and Coordination
The process is highly regulated and coordinated through hormonal and nervous signals. For example, food in the stomach triggers the release of hormones like gastrin, which stimulates the production of gastric acid and pepsinogen. Similarly, when chyme (partially digested food) enters the small intestine, it prompts the release of hormones like secretin and cholecystokinin (CCK), which stimulate the pancreas to release its digestive enzymes and the gallbladder to release bile, aiding in fat digestion.
This complex system ensures that enzymes are released at the right time and in the correct sequence, optimizing the efficiency of digestion and nutrient absorption.
In understanding the workings of enzymes, we uncover the intricacies of life’s biochemical underpinnings, highlighting the exquisite design behind biological reactions. Their story is a testament to nature’s ingenuity, offering insights into the marvels of evolution and the potential for scientific advancements. As we close this chapter, let us marvel at the complexity and efficiency of these molecular machines, which ensure the seamless and harmonious continuity of life’s processes.
Learn more:
How Do Enzymes Lower Activation Energy?
How Does pH Affect Enzyme Activity?
References:
Stryer L, Berg JM, Tymoczko JL. Biochemistry (5th ed.). San Francisco: W.H. Freeman.
Murphy JM, Farhan H, Eyers PA. “Bio-Zombie: the rise of pseudoenzymes in biology.” Biochem Soc Trans.
Radzicka A, Wolfenden. “A proficient enzyme.” Science. 267 (5194): 90–931.
Callahan BP, Miller BG. “OMP decarboxylase—An enigma persists.” Bioorganic Chemistry.
Williams HS. A History of Science: in Five Volumes. Volume IV: Modern Development of the Chemical and Biological Sciences. Harper and Brothers.
Table of Contents
