Tesla pioneered our greatest hope in this space with the development and popularization of battery technology. The demand for lithium-ion battery technology is growing faster than the supply of lithium can satisfy.
So, we need a multi-faceted approach to solve this problem. Another solution, the favorite industry to take over from fossil fuels, is hydrogen/water fuel technology not long ago. Toyota and Shell are working to develop this industry.
Hydrogen has three primary obstacles to overcome to become a viable energy source for any industry: Safety, infrastructure, and cost. If water fuel cells make it to public roads at scale, the hydrogen must be perceived as safe.
How to make a saltwater fuel cell? The production process of hydrogen is pretty simple. It uses a technique called electrolysis to separate water into hydrogen and oxygen. The electrolyzer consists of two metal-coated electrodes and a DC power source, which provides a negative and positive charge. The rate of production of oxygen and hydrogen depends on the electric current.
But pure water is not very conductive. We need to increase the voltage or conductivity to achieve adequate hydrogen production. It’s much more efficient to increase conductivity. So an electrolyte in salt is often included as a charge carrier. It is the oldest and most well-established production method for hydrogen.
How to make an engine run on water as fuel? (Water-powered engine)
When we read our automobile book, we commonly think about How water can be used as fuel? Or, How can water be turned into fuel? Also, it’s a general question we find in our exam papers. So let’s know about it.
Hydrogen Fuel System: The hydrogen generated by the electrolysis device is then channeled into the car’s engine, where it mixes with air. When this mixture is ignited, a mini explosion drives the engine.
Water and Power Supply: The car would need water for the electrolysis device and a power source capable of supplying the necessary electricity for electrolysis. This could come from the car’s alternator, but it would significantly load the vehicle’s electrical system.
Electrolysis Device: Install a device in your car that can perform electrolysis. This device would use electricity (usually from the car’s battery) to break down water into hydrogen and oxygen.
Electrolysis is splitting water into hydrogen and oxygen by an electric current. This current is created by placing two electrodes in the water. These electrodes are made of different metals. So one is an anode, and the other is a cathode. The cathode is where the electric current flows from, and the anode is where it flows to. One is considered positive, while the other is negative.
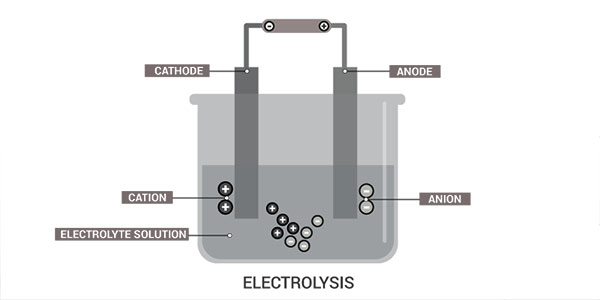
If you’ve ever taken a chemistry class, you might know that water is H2O, which means it’s two hydrogen atoms and one oxygen. But these atoms are covalently bonded, which means they share electrons. When electricity splits that bond, oxygen gets a little sneaky and steals an electron from the hydrogen. So then, these molecules become charged and become attracted by either the cathode or the anode.
Pure water is a pretty poor conductor of electricity. So an electrolyte is added to the water like lithium or sodium. Electrolytes have more ions that allow the electrical energy to move through them. When water is broken up, the oxygen might bind with the sodium, but the hydrogen bonds, forming hydrogen gas.
But why would you do this? What’s the point besides having fun in chemistry class? Well, to fuel, it’s a pretty explosive element. So it could be an excellent fuel source.
Why is water not a good fuel? (Scientific explanation)
Have you heard of hydrogen fuel cells? Hydrogen is in a lot of things. It’s everywhere. About 75% of the mass in the universe is hydrogen. That’s a lot, but hydrogen is a bit energetic molecule. It likes binding with other molecules. So if you want to get hydrogen by itself, you’ve got to be a small home wrecker, get all up in that, and break it up sometimes by using electricity.
You can store that hydrogen once you break up water and use it as fuel. Michael Faraday first discovered the phenomenon of electrolysis back in 1834. So why hasn’t it been a thing in the past century? It’s those dang electrodes.
Many materials corrode when the current is applied, so they don’t last long. So you want an anode material that is resistant to damage from oxidation and a cathode material that is resistant to damage from reduction. But both need to be capable of the appropriate reaction.
What is the future of hydrogen fuel/water-powered?
Scientists are working on it. One study published in Advanced Materials used a cobalt coating that worked for both. The researchers found that it’s cheaper than other materials and even scalable. They think it could even be scaled up to a huge industrial size.
Imagine whole towns being powered by it! Besides powering towns, electrolysis could power robots. Some might use it to power a giant robotic eel that could swim the oceans of Jupiter’s moon Europa.
So it seems like this technology is well on its way to becoming a huge thing, which is excellent because it could be a key to a greener tomorrow. On that note, Toyota is fueling the future with a new Toyota Mirai using hydrogen’s most abundant element in the universe. The Numerai is looking to the future with sustainability in mind.
Read more similar topics:
Why Do We Not Use Water-Powered Cars?
Is It Possible To Use Water As Fuel In The Future?
What Happens To Nuclear Waste?
References:
Edwards, Tony. “End of the road for a car that ran on water.”
State of New Jersey Department of Law and Public Safety, Wayback Machine.
Lopez, Allison. “Inventor, 82, gets 20 years for ‘estafa'”. Philippine Daily Inquirer.
“The Truth About Water-Powered Cars: Mechanic’s Diary.” Popular Mechanics.
Professor doubts water car claims – A leading alternative fuels expert throws water on a Japanese company, claiming it’s developed the world’s first car powered by just water. Professor Theodosios Korakianitis at the Queen Mary University of London says water alone is insufficient to get your car going.
